Alzheimer’s Disease: Comparing FDA Approved Medications

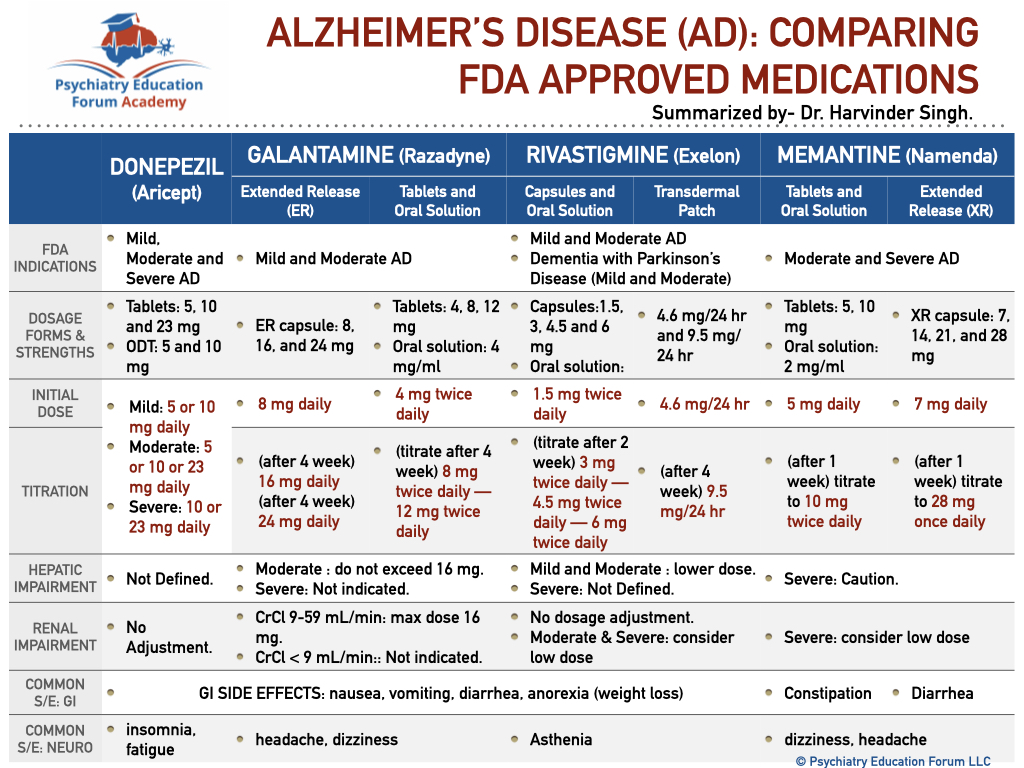
Alzheimer’s disease is the most common type/cause of dementia. As we know that FDA recently approved Aducanumab (Aduhelm) for the treatment of Alzheimer’s Disease (read detailed post here). This post will compare the following old FDA approved medications for Alzheimer’s disease:
- Donepezil
- Galantamine
- Rivastigmine
- Memantine
These medications are compared in the following sections:
- FDA Indications (severity of dementia)
- Dosage forms & strengths
- Initial dose
- Titration
- Hepatic impairment
- Renal impairment
- Common GI side effects
- Common Neurological side effects
SUMMARY:
FOR PEFA MEMBERS:

This PEFA academy chapter will compare these FDA-approved medications in a following 40-minutes video discussion. In addition to the above 8 topics/sections, following topics are also discussed:
- Switching Galantamine from tablets/oral solutions to extended-release formulations.
- Switching Rivastigmine from tablets/oral solutions to transdermal patch formulations.
- Switching Memantine from tablets to extended-release formulations.
INTERESTED IN LEARNING MORE?
JOIN PSYCHIATRY EDUCATION FORUM ACADEMY MEMBERSHIP
This is a closed membership for medical professionals only.
- 30+ Courses/Sections: Each chapter within these sections is of direct clinical relevance for your daily practice.
- Journal Club: we will post the most recently published psychiatry articles relevant to your daily clinical practice.
- Coffee Club: contain short discussions with clinical experts in the field of psychiatry.
- Essentials of Inpatient Psychiatry Book: All chapters from this book are included in the academy sections.
- Discussion Forum & Community: Connect with other medical professionals and discuss your difficult-to-treat clinical cases.
- Conference Discounts: Academy members get discounted access at our conferences.
- Goal: is to have all important clinically relevant topics in one place for ease of access.
SUBSCRIBE TO OUR EMAIL NEWSLETTER:








