Aducanumab: New Medication FDA Approved for Alzheimer’s Disease


June 07, 2021
Today FDA has approved Aducanumab (Aduhelm) for the treatment of Alzheimer’s Disease. This is the first novel treatment to target the underlying pathophysiology of Alzheimer’s Disease and thereby slowing the disease decline. FDA has approved this medication using an accelerated approval pathway.
This post will summarize Aducanumab (Aduhelm) in the following sections:
- Mechanism of Action.
- Dosage & Administration.
- Summary of Controversial Phase 3 Clinical trials: EMERGE & ENGAGE.
- FDA Press Release.
- Adverse Events.
(1) Mechanism of Action:
- Human-derived antibody targeting amyloid-β (Aβ)
- Selectivity for aggregated forms of Aβ plaques & causing reduction of these plaques in the brain.
- Derived from B-cells collected from elderly subjects who appear phenotypically resistant to cognitive deterioration.
- Aducanumab discriminates between monomers and oligomeric or fibrillar aggregates based on weak monovalent affinity, fast binding kinetics and strong avidity for epitope-rich aggregates.
(2) Aducanumab (Aduhelm) Dosage & Administration:
Available Dose: Injection:
- 170 mg/1.7 mL (100 mg/mL) solution in a single-dose vial
- 300 mg/3 mL (100 mg/mL) solution in a single-dose vial
Recommended Maintenance Dosage:
- 10 mg/kg administered as an intravenous infusion over approximately one hour every four weeks.
- Dilution in 100 mL of 0.9% Sodium Chloride Injection, USP, is required prior to administration.
- Administer as an intravenous infusion over approximately one hour via a 0.2 or 0.22 micron in-line filter.
Instructions Before Initiating Treatment:
- Obtain a recent (within one year) brain MRI prior to initiating treatment.
- Obtain MRIs prior to the 7th and 12th infusions.
- If radiographic severe ARIA-H is observed, treatment may be continued with caution only after a clinical evaluation and a follow-up MRI demonstrates radiographic stabilization (i.e., no increase in size or number of ARIA-H).
(3) Summary of Controversial Phase 3 Clinical Trials: EMERGE & ENGAGE
Aducanumab (Aduhelm) approval is considered controversial by some due to it’s two phase 3 clinical trials with mixed results:
TRIAL #1: EMERGE: 221AD302 Phase 3 Study of Aducanumab (BIIB037) in Early Alzheimer’s Disease
- Initially, this was considered a failed study & was terminated (study was discontinued based on futility analysis done and not based on safety concerns).
- Later with the availability of additional blinded data, the prespecified analysis of the primary outcome—Clinical Dementia Rating Sum of Boxes (CDR-SB)—showed that the EMERGE trial met its primary outcome (which is Change from baseline in CDR-SB score)
- Significant 22% slowing of decline on the CDR-sb.
SECONDARY OUTCOME MEASURES:
- All secondary measures including the Mini-Mental State Examination, Alzheimer’s Disease Assessment Scale-cognitive subscale, and the Alzheimer’s Disease Cooperative Study Activities of Daily Living (ADCS ADL) scale showed statistically significant drug-placebo differences.
- The ADCS-ADL scale showed a robust 40% slowing of functional decline in the treatment group compared to the placebo.
- Neuropsychiatric Inventory (that assesses an array of behavioral changes common in Alzheimer’s Disease) showed an 87% reduction from baseline scores in the high dose group of EMERGE.
- 84% reduction in caregiver distress seen.
TRIAL #2: ENGAGE: 221AD301 Phase 3 Study of Aducanumab (BIIB037) in Early Alzheimer’s Disease
- Initially, this was also considered a failed study & was terminated (study was discontinued based on futility analysis done and not based on safety concerns).
- Later even with the availability of additional blinded data, ENGAGE trial did not met its primary outcome.
- Note that benefits were observed in ENGAGE participants who were treated with the high dose for longer periods.
NOTE ==> Amyloid plaques measured by amyloid positron emission tomography (PET) were markedly decreased by aducanumab in both trials:
EMERGE TRIAL:
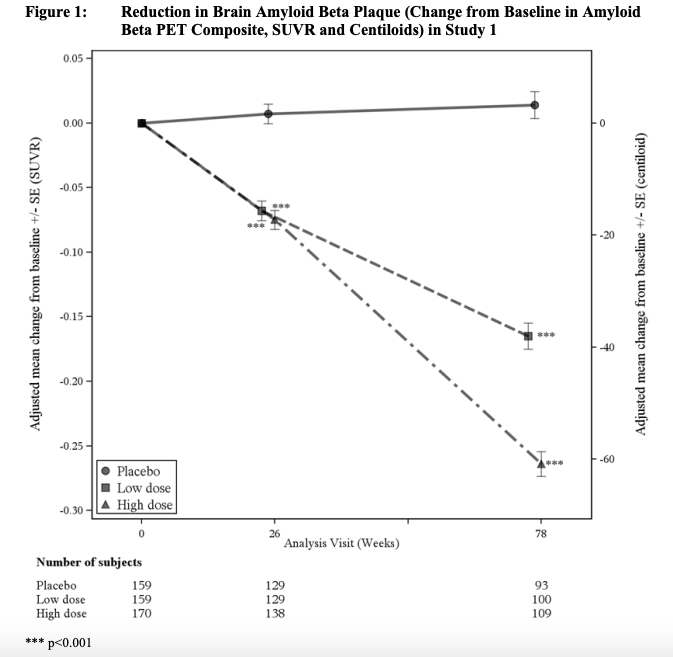
ENGAGE TRIAL:
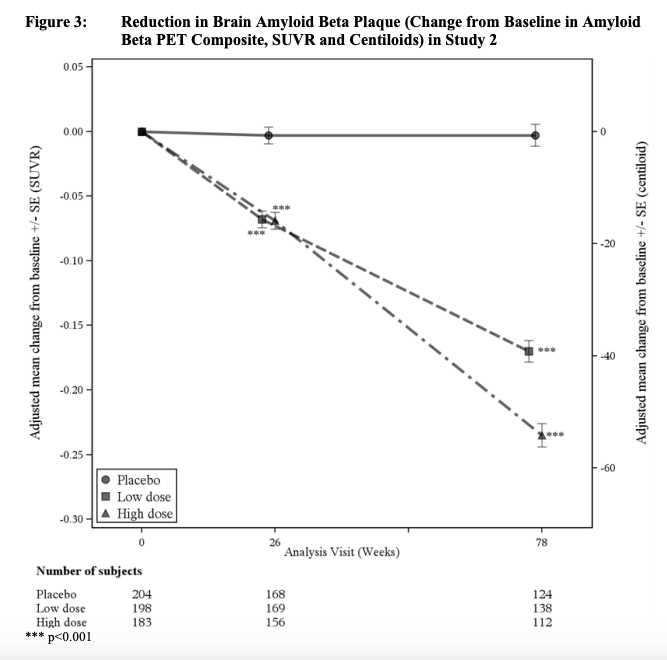
(4) According to FDA Press Release:
“FDA is requiring the company, Biogen, to conduct a new randomized, controlled clinical trial to verify the drug’s clinical benefit. If the trial fails to verify clinical benefit, the FDA may initiate proceedings to withdraw approval of the drug.”
(5) Adverse Events Seen in Clinical Trials:
Common Adverse Events:
- ARIA (amyloid-related imaging abnormalities): mostly asymptomatic but can cause headache, confusion, dizziness, vision changes, or nausea.
- Headache
- Fall
- Diarrhea
- Confusion/delirium/altered mental status/disorientation.
Serious Adverse Events/Warning:
- Hypersensitivity reactions, including angioedema and urticaria
REFERENCES:
- FDA Approval statement (FDA website)
- FDA Press Release (FDA)
- Prescribing Information (PDF)
- Cummings J, Aisen P, Lemere C, Atri A, Sabbagh M, Salloway S. Aducanumab produced a clinically meaningful benefit in association with amyloid lowering. Alzheimer’s Res Ther. 2021 May 10;13(1):98. PMID: 33971962; PMCID: PMC8111757. (Pubmed)
- FDA: Aducanumab for the Treatment of Alzheimer’s Disease: Clinical Overview of Efficacy. (PDF)
- Arndt JW, Qian F, Smith BA, et al. Structural and kinetic basis for the selectivity of aducanumab for aggregated forms of amyloid-β. Sci Rep. 2018;8(1):6412. (Pubmed)
- 221AD302 Phase 3 Study of Aducanumab (BIIB037) in Early Alzheimer’s Disease (EMERGE)
- 221AD301 Phase 3 Study of Aducanumab (BIIB037) in Early Alzheimer’s Disease (ENGAGE)
SUMMARIZED BY:
INTERESTED IN LEARNING MORE?
JOIN PSYCHIATRY EDUCATION FORUM ACADEMY MEMBERSHIP
This is a closed membership for medical professionals only.
- 30+ Courses/Sections: Each chapter within these sections is of direct clinical relevance for your daily practice.
- Journal Club: we will post the most recently published psychiatry articles relevant to your daily clinical practice.
- Coffee Club: contain short discussions with clinical experts in the field of psychiatry.
- Essentials of Inpatient Psychiatry Book: All chapters from this book are included in the academy sections.
- Discussion Forum & Community: Connect with other medical professionals and discuss your difficult-to-treat clinical cases.
- Conference Discounts: Academy members get discounted access at our conferences.
- Goal: is to have all important clinically relevant topics in one place for ease of access.
SUBSCRIBE TO OUR EMAIL NEWSLETTER:









